Sat Jan 31 15:47:09 PST 2009
Vitamin-C - A Food Preservative Essential For Life
Do you reach for a bottle of vitamin-C tablets when you feel a cold coming on? Vitamin-C, or ascorbic acid, is reputed to have the ability to strengthen the immune system and improve our ability to fight back against invading influenza viruses. Hence, many people dose themselves with vitamin-C tablets, or citrus fruit, at the onset of a cold.
Medical trials of vitamin-C's effect on colds, both as a treatment, and as a preventative have shown only modest value. On average, taking a vitamin-C supplement may reduce the number of days of cold which one person suffers per year by around 10 percent. This is much less than a day of relief for most people from a typical cold. And the results are very variable with the most positive results coming from those who may be bordering on vitamin deficiency. So vitamin-C as a treatment or preventative measure for colds has only limited value.
However, vitamin-C is certainly an effective treatment for vitamin-C deficiency and vitamin-C deficiency, or scurvy, produces extremely undesirable effects. The lack of vitamin-C reduces the ability of the body to synthesize collagen, resulting in a range of skin problems including weakened gums, spots, and bleeding from mucous membranes.
Humans cannot synthesize vitamin-C, and a diet that lacks vitamin-C will result in scurvy. The recognition of this fact, and the resulting compulsory eating of citrus fruit for British sailors, resulted in the use of the general expression 'Limey' in America for a British person. (One imagines that the lime colored skin of the British sailors may have strengthened the use of the term...)
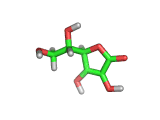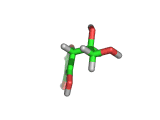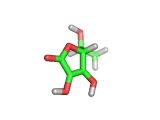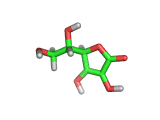
From a chemical perspective the ascorbic acid molecule (which is shown above) is effective at absorbing free radicals and allowing itself to be oxidized in preference to surrounding molecules. It has three oxygen atoms close together attached to its ring structure, (these are shown in red in the model above), and these oxygen atoms can readily donate protons to passing molecules or absorb electrons from passing free radicals.
Consequently, ascorbic acid is often added to foods as a preservative. The fact that it is also a vitamin, which our bodies cannot synthesize, makes its use as a food preservative acceptable.