Wed Sep 17 19:43:30 PDT 2008
BisPhenol-A, BPA
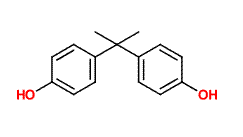
The molecule shown above is bisphenol-a, also known as BPA. Like TATP (triacetone triperoxide) the bisphenol-a molecule is synthesized by allowing acetone to react with other molecules. In the case of bisphenol-a the other molecules are phenol. When catalyzed by an acid (as is also the case in the TATP reaction), two phenol molecules react with one acetone molecule, eliminating water and creating bisphenol-a. The 'a' in this molecule's name is simply taken from 'acetone' showing just how unsystematic common chemical names can be.
Bisphenol-a is used as an antioxidant for the plasticizers in many plastic and polymer products and also inhibits additional polymerization, which leads to brittleness in some plastics. This compound is made in large quantities by the chemical industry and is used in many of the products in your home as a polymer additive.
With its two aromatic rings with hydroxyl groups, bisphenol-a is able to act as an antioxidant by combining with oxygen containing radicals which would otherwise attack the carbon backbone of the plastic. Once the bisphenol-a reacts with the radical, the radical electron is delocalized and stabilized by the aromatic rings of the bisphenol-a molecule and this stops what might otherwise be a chain reaction. The ability of bisphenol-a to react with radicals in this way is a similar to that of vitamin E, which is occasionally added to foods to prevent free radical induced spoiling.
Bisphenol-a has been used in plastics which come into contact with food for many years and while the small quantities of bisphenol-a that reach humans as a result are tiny there is some doubt as to whether bisphenol-a is an entirely safe additive. It has to be said that many molecules reach us from many sources, and even vitamin E should not be taken in substantial doses. So while the concerns about bisphenol-a should not be dismissed, and should be carefully researched, they are also not something to worry about unnecessarily.